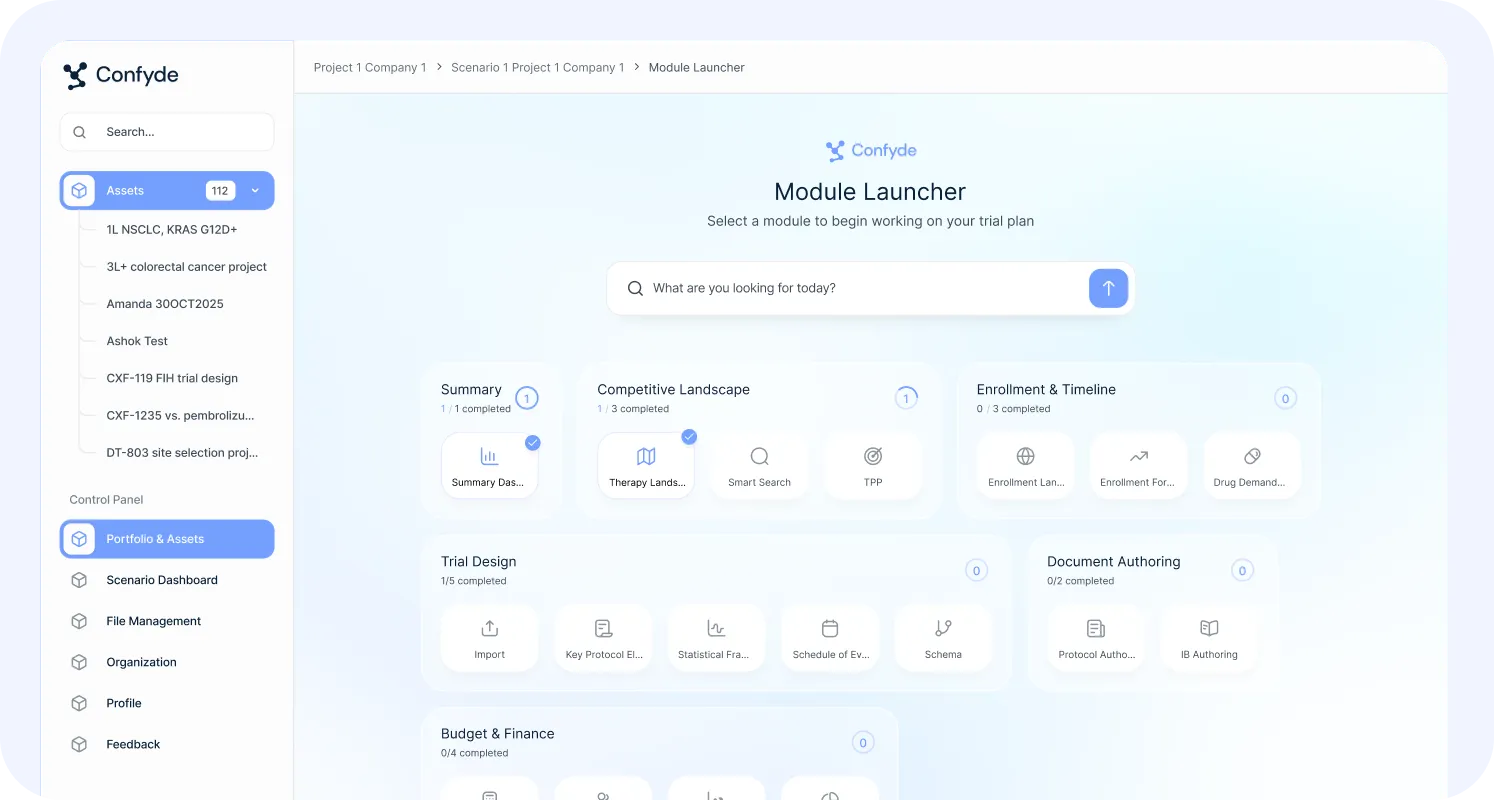
Study Designer
The Study Designer module serves as the structural foundation for downstream analysis and content generation across Confyde.ai.
Users can rapidly construct core study design frameworks by either:

Uploading existing materials (e.g., study synopses, protocols, concepts)

Importing trial data via API integrations (e.g., ClinicalTrials.gov)
Confyde.ai then extracts, organizes, and normalizes key design elements, including:

Population definitions

Endpoints and objectives

Treatment arms and comparators

Eligibility criteria

Study architecture
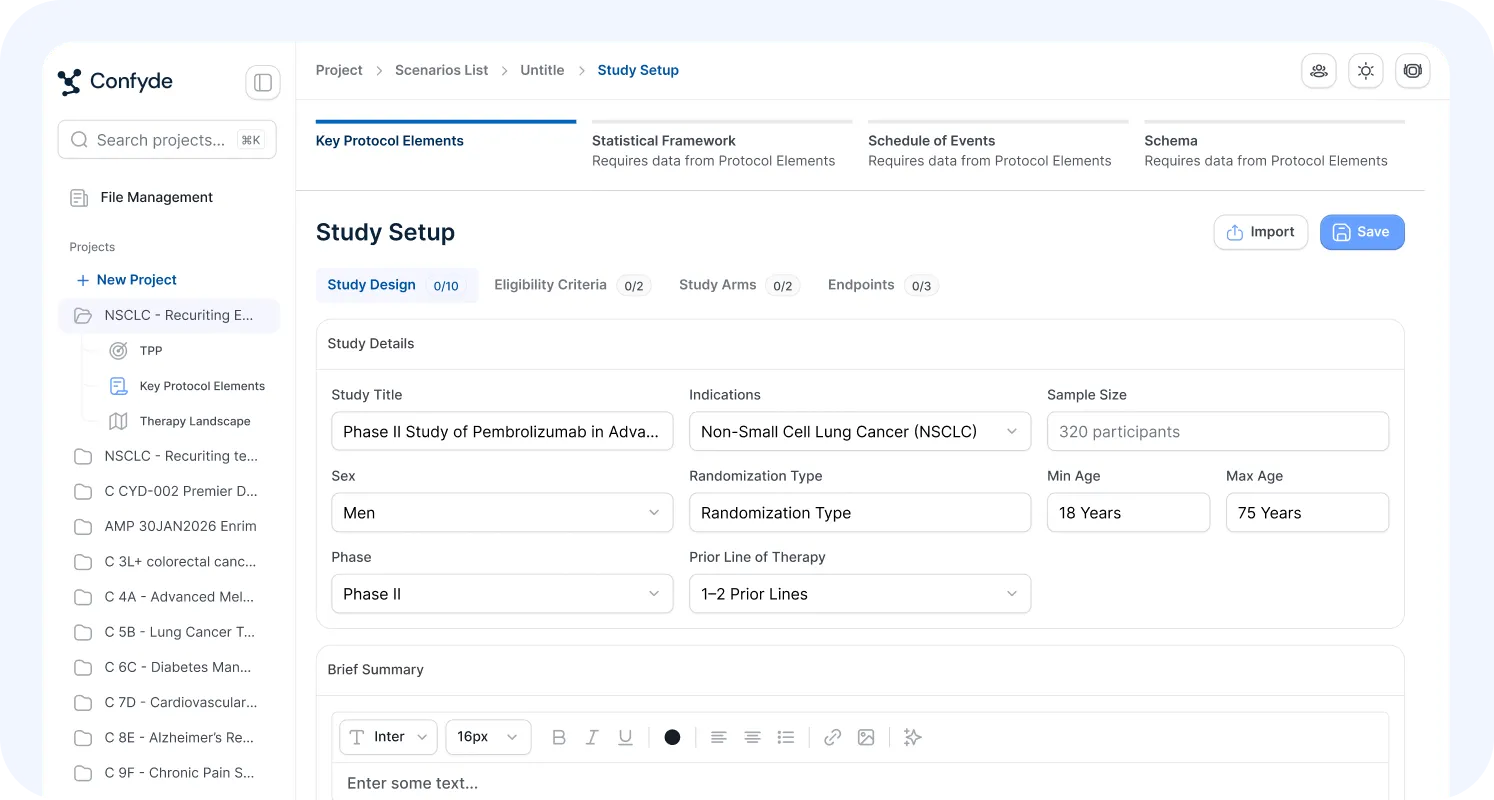
These structured inputs become reusable intelligence assets that inform other modules across the Confyde.ai platform.
Rather than repeatedly redefining study parameters across workflows, Study Designer creates a consistent, development-aware framework that drives coherence, efficiency, and analytical continuity.
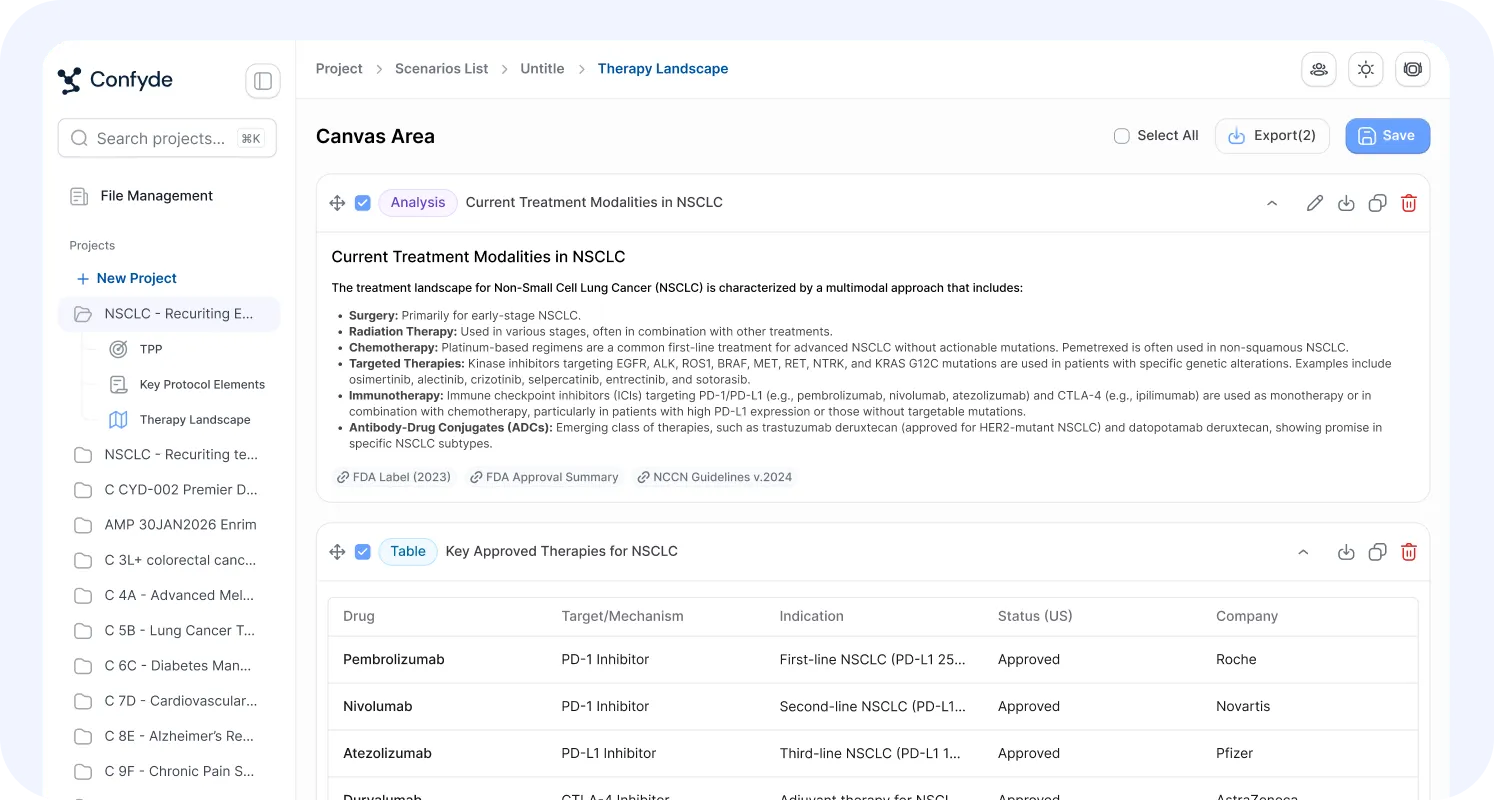
Therapy Landscape
Confyde.ai transforms fragmented clinical and competitive data into structured strategic insight.
This module synthesizes information across trial registries, scientific literature, and development programs to provide a continuously updated view of the therapeutic landscape.
Users can:

Map active and planned competitor trials

Identify positioning risks and opportunities

Analyze evolving standards of care

Detect strategic blind spots

Create and review full-length AI-generated diligence reports
The result is not just data aggregation, but decision-relevant competitive clarity.
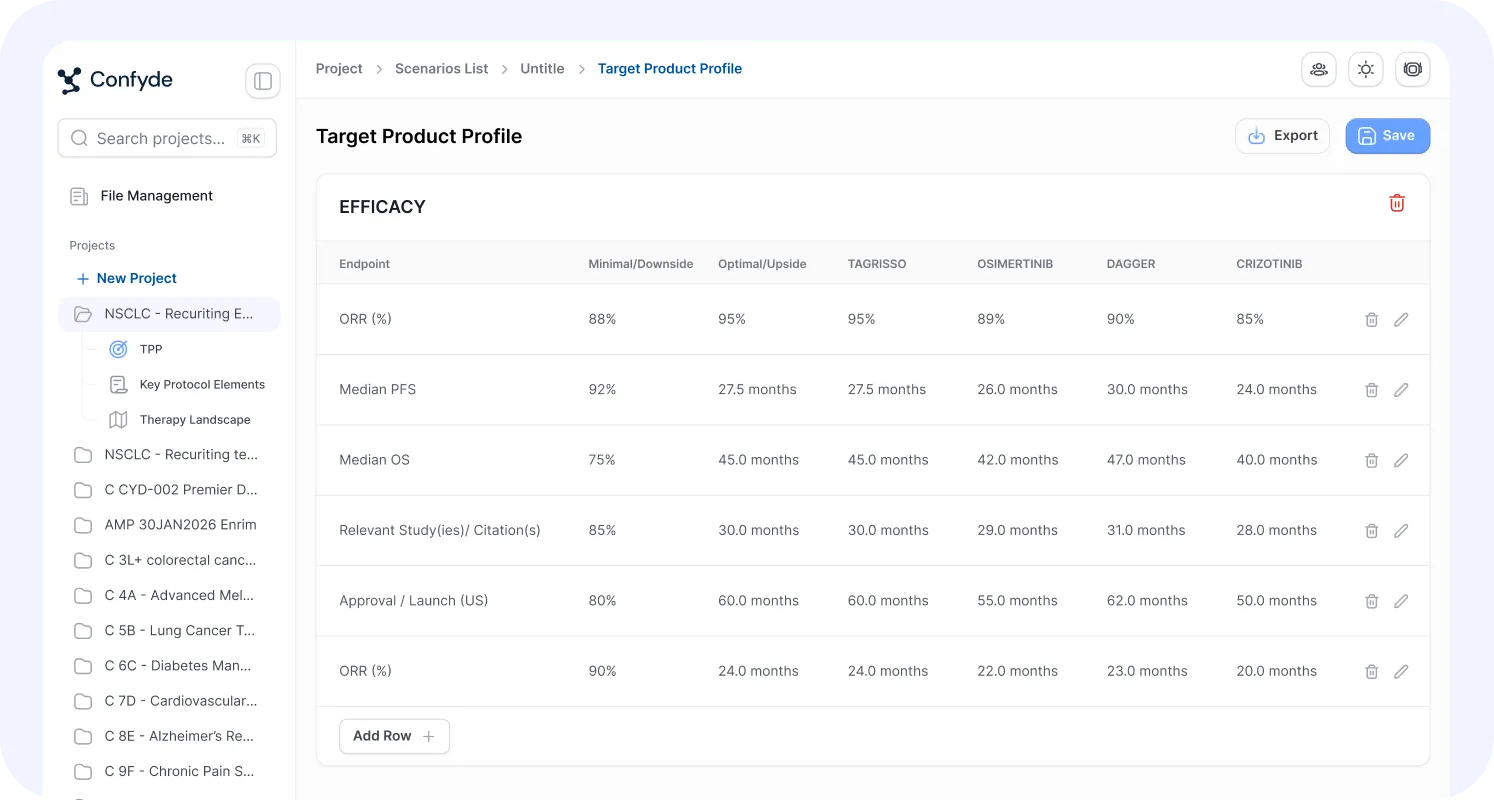
TPP Generator
The Target Product Profile (TPP) Generator helps teams define, stress-test, and refine asset strategy.
By integrating clinical, regulatory, and commercial considerations, this module supports structured articulation of product goals and differentiation hypotheses.
Users can:

Develop draft TPP frameworks

Align endpoints with regulatory expectations

Evaluate competitive differentiation

Iterate on strategic assumptions
This enables earlier, more coherent alignment across development functions.
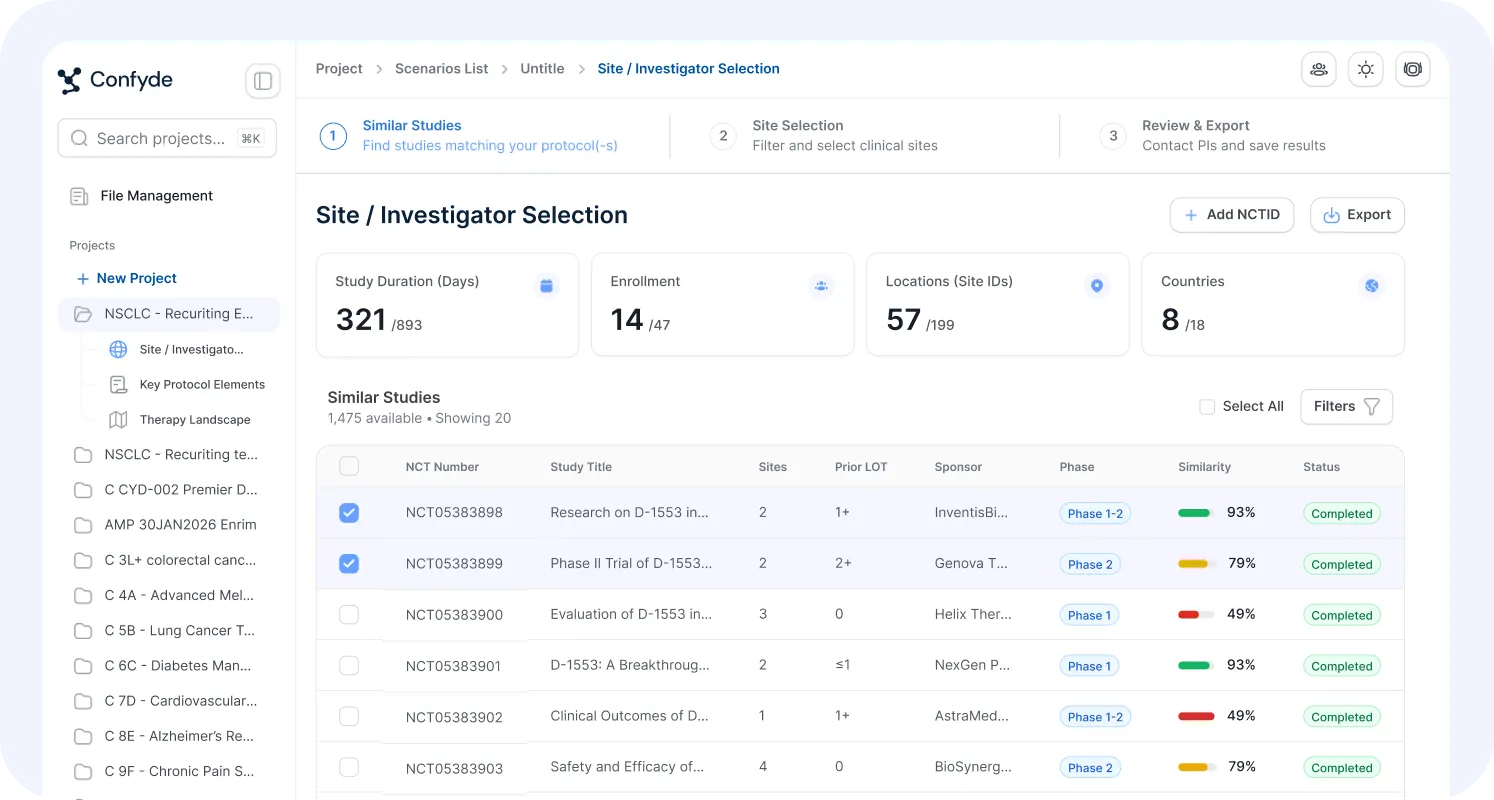
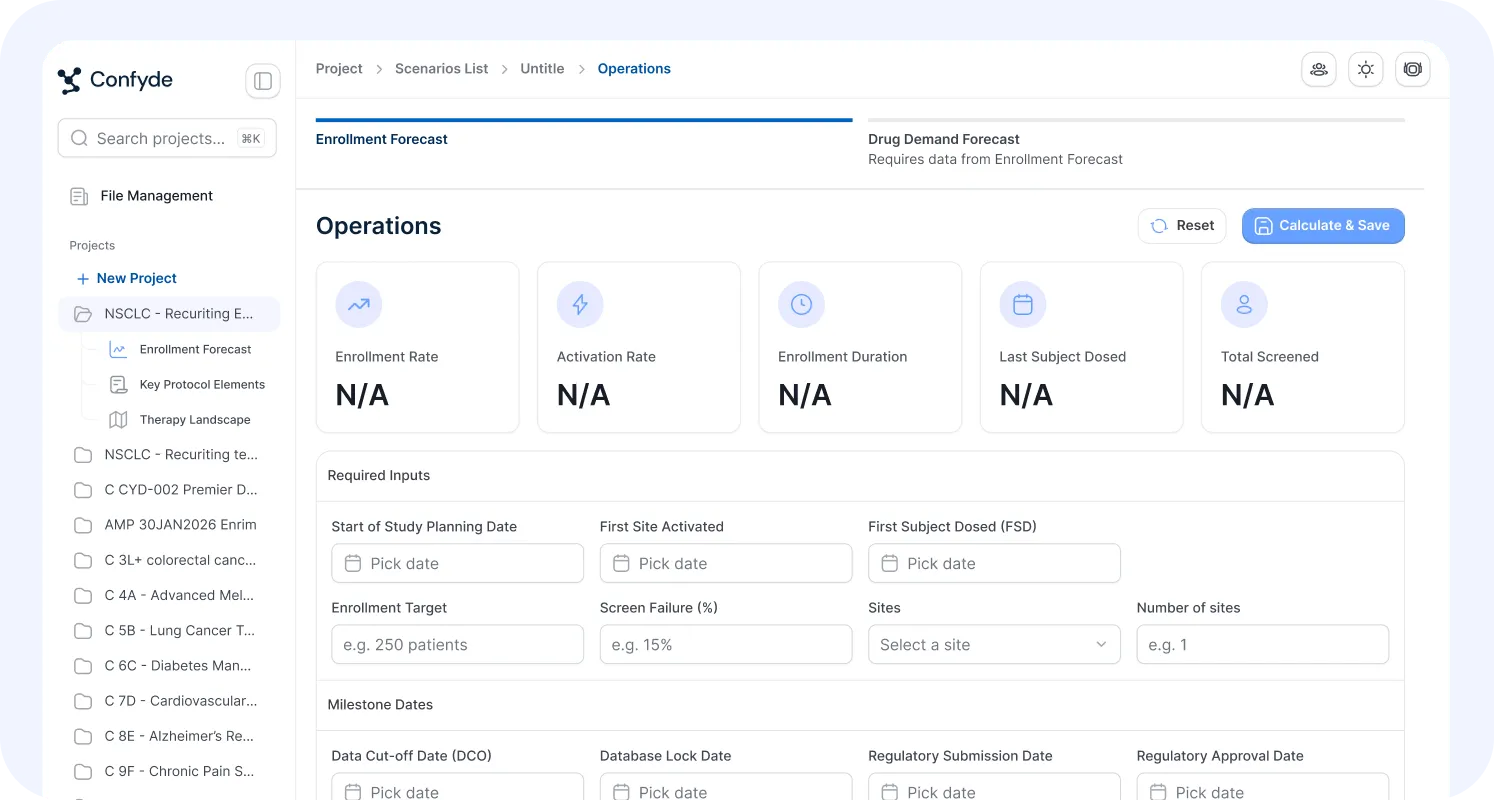
Site Selection/Enrollment Forecasting
Operational decisions can determine trial success or failure.
This module provides data-driven modeling of site strategy and enrollment dynamics, helping teams evaluate feasibility, timelines, and execution risk.
Users can:

Identify optimal site profiles

Forecast enrollment scenarios

Model activation and recruitment timelines

Evaluate geographic strategy tradeoffs
Confyde.ai helps replace intuition-driven planning with quantitative operational intelligence.
Resource/Revenue Forecasting and PTRS
The Regulatory Review module provides structured, evidence-anchored evaluation of development strategy and trial design.
Built around regulatory precedent and agency guidance, it helps teams identify strengths, vulnerabilities, and alignment risks.
Users can:

Assess protocol design considerations

Evaluate endpoint strategy

Identify regulatory risk factors

Explore preferred/alternative pathways
Outputs emphasize traceability, defensibility, and regulatory relevance
Document Authoring
Confyde.ai accelerates the creation and refinement of development documents while preserving scientific and regulatory rigor
Rather than generating generic text, this module operates within a development-aware framework.
Users can:

Draft protocols and briefing materials

Refine existing documents

Maintain structural consistency

Integrate evidence-linked content
The goal is not automation alone, but high-quality, submission-ready documentation.
Ready to transform your clinical trials?
Design higher-probability trials, accelerate execution and prioritise the right assets with Confyde’s AI-native platform.

.webp)
